Legume laboratory discusses efforts to express and localize algal Carbon Concentration Mechanisms (CCM) in Arabadopsis and Tobacco
Legume laboratory discusses a recent effort to introduce an algal carbon concentration mechanism into higher plants for improved photosynthetic efficiency.
Source: Fast-Tracked Photosynthesis
We have previously written on the topic of increasing the efficiency of photosynthesis as a possible method of increasing crop growth and yield. By swapping out the predominant C3 photosynthesis system with the superior C4 system, it was estimated that rice and wheat yields could increase by 50%.
The Long Now Foundation recently published online a talk titled “Radical Ag: C4 and beyond” in which Jane Langdale of the Langdale Lab discusses the 60 odd plant species that have naturally evolved C4 photosynthesis and the efforts to use this knowledge to get C4 photosynthesis into rice.
A different method of supercharging photosynthesis?
And a recent paper in the Plant Biotechnology Journal (which is open access – yay!) investigated the possibility of using a carbon-concentrating mechanism (CCM) commonly found in a number of photosynthetic organisms like cyanobacteria and green algae.
Whilst C3 photosynthesis relies on CO2 passively diffusing to the chloroplasts, resulting in the enzyme which assimilates the carbon into the plant (RuBisCO) not being saturated with CO2, the microbial CCM increases the concentration of CO2 to RuBisCO and thereby increases photosynthetic efficiency.
In algae, the CCM consists of transporters of inorganic carbon at the plasma membrane, chloroplast envelope and carbonic anhydrases. These all work together to deliver the elevated concentration of CO2 to the RuBisCo enzyme.
In this paper, the researchers took a number of steps to check the plausibility of inserting this CCM into tobacco plants:
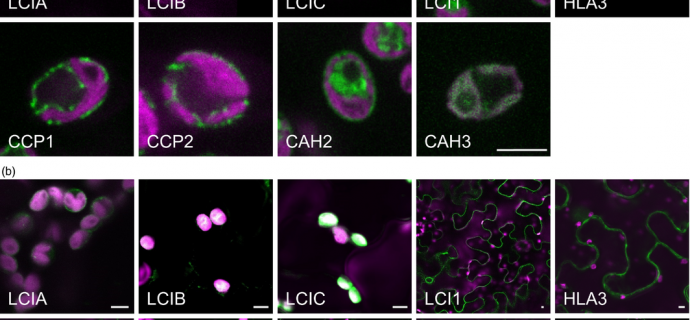
After choosing 10 critical components of the CCM, these components were tagged with fluorescent markers to determine where they were located within Chlamydomonas reinhardtii.
With the usual location of these proteins confirmed, the proteins were expressed in tobacco leaves to determine whether the same components were located in the same cellular locations.
Focusing on two inorganic carbon transporters within this selection of 10 components, the researchers showed that they both function as such when expressed in the outer membrane of a model single-celled organism (Xenopus oocytes) and when transformed into Arabidopsis thaliana.
Finally, they tested the growth of the Arabidopsis plants against wild-type plants of the same species.
The positives
The location of the 10 components were confirmed within the bacteria and 8 of these were located in the same cellular position when expressed in tobacco leaves. 1 protein was mistargeted whilst the researchers struggled to clearly identify the location of another.
Delving further into this issue, the researchers looked for a way to direct one of the proteins specifically to the chloroplast stroma using a specific transporter found in an online plant membrane protein database. Using this transporter and the tagged protein, they were able to demonstrate the ability to re-target a protein from a single celled organism to a specific part of the cell of a higher plant.
Taking two specific proteins for further study, the proteins were expressed in Arabidopsis plants and the location was confirmed as being the same as in the tobacco plants.
The negatives
Actually, the report specifically states that it wasn’t expected that Arabidopsis plants transformed with the two CCM components would have an altered rate of photosynthesis given the complexity of this machinery and the number of components missing. However, they tested and compared growth rates under normal and reduced CO2 concentrations, finding no difference between the transformed and wild-types. They also measured CO2 assimilation rates, again finding no difference.
Conclusion
The paper gives a great insight into complexity involved in demonstrating the plausibility of transforming plants for greater growth rates and, potentially, greater yield from food crops.
The ability to express and redirect proteins is impressive, but also impressive is the catalogue of plant proteins that is continually being built and which can be freely accessed and used for research.
Increasing CO2 sequestration rates is a focal point for many researchers in the quest to grow more food on the agricultural resources we have. Swapping out or supercharging the C3 photosynthesis present in most crops is one method with seeming potential and the detail of the work performed so far gives an exciting insight into the possibility of it coming to fruition.
Source: From The Community: Fast-tracked Photosynthesis | PLOS Synthetic Biology Community











Leave A Reply